2026 Author: Erin Ralphs | ralphs@carsalmanac.com. Last modified: 2025-01-22 21:14:09
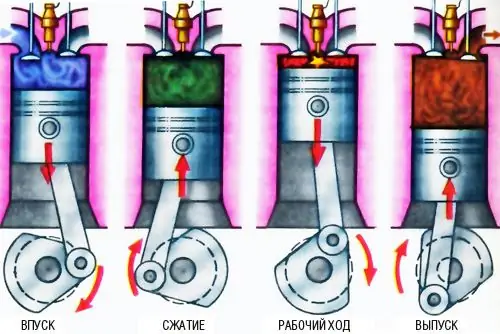
Each engine, regardless of size, type of fuel, power and torque, has a number of technical characteristics that do not change over time. For example, when worn, the engine develops less power than the new torque. In addition, fuel consumption also increases. But there are others, such as piston diameter, stroke, displacement. So, among these values you can find the degree of compression. This is a calculated value.
So, you need to know what the compression ratio is. This is the ratio of the working volume of one cylinder of the engine to the volume of the combustion chamber. Therefore, if the car owner wants to increase the compression ratio, there are two ways to do this: reduce the second (that is, the combustion chamber) or increase the first (that is, the volume of the cylinder). The second way is much more difficult, so tuners prefer to perform all sorts of operations with the cylinder head. This is done by grinding the plate, since the head is one-piece, and the filling method is not suitable here. In addition, in most engines, the distribution of the combustible mixture over the cylinder is calculated, so a violation of the internal geometry is fraught with consequences.

Engine compression ratio affectsmany of its characteristics in everyday use. First of all, this is its torque, since the higher the pressure above the piston, the more energy it receives during the power stroke. As a result, the pressure on the crankshaft journal increases, which means that the torque of the engine also increases.
Another characteristic that is directly affected by the compression ratio is fuel consumption, and this dependence is inversely proportional, that is, the more the first, the less the second. But not every fuel can be used at a high compression ratio. For example, if the degree exceeds 9.0, then gasoline must be with an octane rating of at least 92 (AI-92). The fact is that the low octane number of gasoline indicates its instability to detonation, that is, pre-ignition from pressure and temperature.

This leads to increased wear of the connecting rod and piston group, since the explosion of the mixture occurs even before the piston reaches top dead center. This reduces engine power. In addition, the temperature regime increases, which is fraught with other, even more terrible consequences, such as burning the rings to the cylinders.
The compression ratio of a diesel engine is much higher, sometimes even twice. It reaches 16, since the ignition of the combustible mixture does not occur from the ignition spark, but from the pressure in the combustion chamber. The pistons here have special sleeves in the bottom, which serve to guide the mechanism straight down.
In conclusion, it is worth recalling once again thatis the compression ratio. This is a characteristic of the engine that does not change throughout the entire period of operation, since the dimensions remain the same. Many people confuse compression ratio with engine compression. We will not go into details of what compression is, we will only say that this is pressure, which is measured using a pressure gauge. Our compression ratio can only be calculated. In order to do this, you need to measure the volume of the combustion chamber. This is done by adding liquid from a beaker with a division of 1 ml.
Recommended:
Chevrolet Niva alternative: car description, compliance with the necessary conditions and price / quality ratio

On our roads you need to drive the appropriate cars. Strictly high ground clearance is needed, four-wheel drive is desirable, short overhangs, and it will also be good if the components for the car are inexpensive. And if the car is also comfortable, then this is generally good. All these parameters correspond to the Chevrolet Niva. Today we will only briefly touch on this car, in more detail we will consider the topic of alternatives to the Chevrolet Niva on the market today
The correct ratio: the characteristics of the cargo - the length, width and height of the truck

For any transported goods, a certain type of suitable vehicle equipped with special equipment is usually chosen. According to the volume, weight, temperature regime of storage and the method of packing the cargo, determine the type of semi-trailer
Compression is Engine compression - what is it?

Compression is the determining factor in engine longevity. It is from this indicator that the degree of wear of various parts of the motor depends. Therefore, motorists are trying to support it in various ways. Engine compression - what is it and how is it measured? In today's article, we will try to describe this phenomenon in detail, and also learn how to measure it with our own hands
Compression ratio and octane number of gasoline

Very often, novice drivers wonder what kind of gasoline is better to fill in a car. At the moment, there are several varieties of fuel, with different octane ratings. Which type is better to use so as not to "sentence" the motor? What is the octane number and compression ratio of an engine? Let's try to understand our today's article
Compression and compression ratio: difference, principle of operation, similarities and differences
Does every vehicle owner clearly understand the difference between compression and compression ratio? Meanwhile, this is by no means the same thing, as some motorists (often beginners) believe, due to little experience. This should be understood at least in order to be able to fix a slight malfunction on your own, without the help of a qualified specialist. In addition, it will be the accumulation of personal experience, which in any case will not hurt

